Closed Loop Oncology from Medication Compounding to Administration and Beyond.
James Waterson. RN, M.Med.Ed. MHEc. Medical Affairs Manager, Medication Management Solutions, Becton Dickinson. Middle East and Africa. James.waterson@bd.com
Lisa Gangol. RN, BSN. Senior Clinical Resource Consultant, Medication Management Solutions, Beckton Dickinson. MMS-MENAT.
Aya A.Kabbani. Pharm.D. Product & Clinical Manager, Medication Management Solutions, Beckton Dickinson. MMS-MENAT.
Larry Neal. Regional Marketing Manager, Medication Management Solutions, Becton Dickinson. Europe, Middle East and Africa.
Potential errors of Wrong Patient, Wrong Medication, Wrong Dose, Wrong Documentation, and Wrong Time can all take place at the bedside, and among all the stages of the medication chain -from prescription through to administration- medication errors which occur at the point of administration are the most prevalent at 38% of all medication errors. [1] With only a 2% chance of detection of error, in terms of Failure Mode Effect Analysis (FMEA) the IV administration process consistently scores as a high-risk activity by virtue of this difficulty of detection, with a Detectability Score of 10 (0 = Minimum Harm Risk – 10 = Maximum Harm Risk) commonly being applied by organizations utilizing FMEA. IV-medication administration is therefore associated with a far greater risk of medication error than oral administration. [2,3]
This is the dangerous ‘gap’ that Connected Compounding Solutions and Interoperability Capable Infusion Pumps can close, as they effectively loop back IV-medication administration directly to the original prescription and ensure completion of the documentation of administration. [4]
Bidirectional-interoperability capable IV pumps achieve this through direct communication with the patient’s Electronic Health Record (EHR):
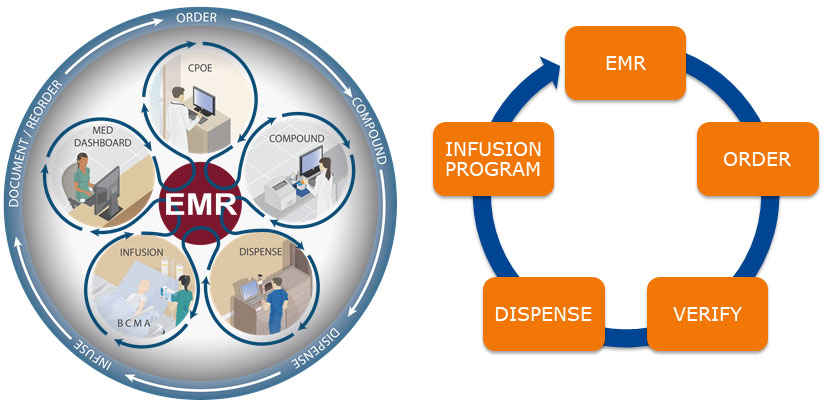
Bidirectional Interoperability is the most advanced form of Barcode Medication Administration: The smart pump directly communicates with the EHR to confirm the prescription is current and correct confirms the patient identity, and auto-documents administration.
The barcode on the IV-medication bag or syringe triggers this communication during a process of scanning the Patient’s ID band, scanning the medication to be given, and scanning the barcoded unique identifier of the IV pump. The pump is then triggered to communicate with the EHR and the confirmation with the Computerized Provider Ordering Entry (CPOE) system allows for auto-population of the pump with an order string that carries the correct dose, duration or rate, correct volume of the IV bag and the patient ID.

The order string barcode applied during compounding allows bedside confirmation of right patient, and the auto-population of the smart pump with the right dose and duration for the oncology regimen medication, the loop is closed by auto-documentation of administration.
This is truly streamlined safety as it can also reduce the time spent programming the pump by over 80%, [5] and oncology care needs streamlining badly. One estimate suggests that by 2040, globally, the number of patients requiring at least first-line chemotherapy each year will have increased from a 2018 baseline by 53% (from 9.8 million to 15 million individuals) [6] In one recent study the annual compounding requirements for oncology IV-medications in a regional cancer care unit increased by over 15 times between 2012 and 2020 [7].
The hospital pharmacy is central to the above EHR interoperability workflow as it is here where the order is electronically received from the CPOE, the medication is compounded and the individual and unique barcode with an order string is created from the CPOE, and the individual patient ID. It is possible to manage this process manually, but many of the critical steps involved in compounding carry potential errors, [8] and it is far better to have integration between the CPOE and the compounding software to avoid breaks in the medication chain, and to reduce the risk of transcription errors. [9] Prescribing software should also include ‘Hard Stops’ for dosing and automation of calculations for Area Under the Curve (AUC) dosing. Ideally, the CPOE gives prescribers access to computer-based standardized protocols, which reduce non-standard regimen creation, can make for a faster regimen build, and automatically calculate doses and create preparation guidance. With integration, physician prescriptions can be received electronically into the pharmacy for verification, and if approved, for push-communication to the compounding unit.
Interoperability between the EHR and IV Pumps is an ideal solution for whole hospital medication safety, as it requires an investment in communication engines to link pumps to the EHR and an organization-wide network capable of carrying these messages, ideally via secure wireless communication. Modern smart pumps can make this a highly protected connection via Advanced Encryption Standard (AES) 128-bit key encryption using Cipher Block Chaining (CBC). This encryption applies to a vast number of connections with modern smart pump servers being able to manage up to 32,000 pump channels with full, and secure, bidirectional interoperability across hospital campuses with multiple sites. Updating Dose Error Reduction Software (DERS) libraries on pumps to match central formulary changes with new medications and oncology regimens is vital to ensuring the presence of a second FMEA medication safety gate. Updating DERS libraries rapidly and safely across large oncology facilities and ambulatory care units can only really be achieved when there is the capability to deploy the new library to connected pumps via extensive wired or wireless networks. [4] Auto-population of infusion orders to connected pumps facilitates the utilization of DERS, with all scanned orders utilizing the customized drug library when programmed, verified, and started.
A large capacity DERS smart pump library of holding at least 3,000 to 10,000 medication entries is also vital as we see more and more novel drug treatments, an expanding array of Monoclonal Antibodies (MAB) [10], and a need for multiple protocols for individual medications becoming standard practice. [11]
As mentioned above efficient oncology compounding software is a central part of any EHR interoperability solution for IV-medications, as every prescription must pass through the pharmacy for reconstitution and application of the unique patient-medication matched barcodes. The pharmacy compounding unit is truly the engine house of IV-medication production and of IV-medication safety.
A compounding workflow software solution can undertake both preparation and documentation assistance and checks. A software suite should support initial pharmacist verification of the order, gravimetric checks, and incorporate a monitor inside the work area with a minimal interaction interface with step-by-step instructions. The software can propose a list of items that includes the minimum medication vials, diluents and consumables required to prepare the dose. Scanning of individual components ensures a recipe match for all items including the final IV administration bag. The system must also carry ‘Hard Stops’ for dosing out of tolerance (±4% tolerance is accepted by most institutions). [12] The compounding system should only deliver a patient-medication label after all the steps of compounding are successfully complete, which makes it ideal for building into an FMEA process, as the steps involving risk are all before the final verification checks and issuing of the label with a unique preparation and patient identification number. [7,8] The software must also be capable of recording all cancelled mixes, tolerance limit breaches, incorrect item scans, and the resolutions of alerts by the user. Date-time stamps are automatically applied to all these alerts and actions by modern compounding solutions. This data is ideally stored locally on an MS-SQL database inside the hospital firewall. Documentation in the central pharmacy of compounding statistics for each preparation can be aggregated and this can assist with forecasting.
Viewers and dashboards of the entire IV-medication management process from prescription to administration help senior pharmacy managers to identify bottlenecks in the pharmacy fulfilment process. Compounding units can also view infusion status in all nursing units, allowing for just-in-time resupply of critical medications and prioritizing of activity and queuing of compounding requests. New medication dose requests are easily identifiable in such an arrangement, and such systems are extendable into inventory control and forecasting functions.
Tracking of medications and the ability to reintroduce unused but highly valuable oncology medications back into the system can be a real challenge. Medication stock tracking can include integration between pharmacy applications and Enterprise Resource Planning (ERP) systems for ‘stock-in stock-out’ reconciliation. Of course, once the oncology product is compounded there can be an issue of the storage of bulky and high-volume infusions requiring refrigeration with issues of track and control as, commonly, the refrigerators used in facilities are not connected to the pharmacy system. Full integration of infusion pumps allows the central pharmacy to view dashboards of each patient’s current infusion, and the time at which it will end. This can help with preparation of the next oncology medication for the patient’s regimen to be delivered Just In Time (JIT).
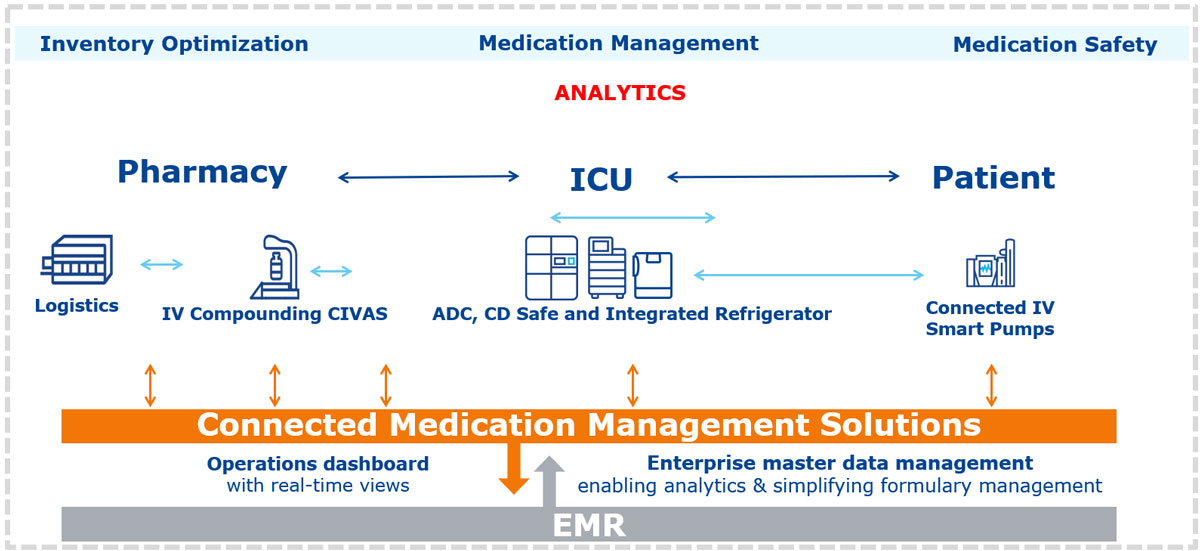
Integrating infusion pumps and automated dispensing cabinets allows for more accurate forecasting of oncology medication needs as infusion rates can be viewed in real-time and overall medication consumption can be assessed and shared across facilities.
Aggregation of this data and integration with par-level reporting data from Automated Dispensing Cabinets (ADC) for oral medications and networked and integrated ADC style medication fridges for IV-preparations also allows for more accurate forecasting going forward. This data can also theoretically feed directly into regional and national systems, many of which currently rely on individual facilities regularly reporting critical oncology medication stock-levels. These are currently often calculated from weighted averages, and projected needs based on historical order volumes. National or regional coordination of large-scale oncology strategies needs a dynamic real-time data-feed to allow for centralized ordering and distribution and inter-facility ‘lending’ arrangements for critical need. Sharing of scarce medications during shortage-crises can be a central part of our strategy to streamlining services with the goal of increasing cancer survival rates across healthcare systems.
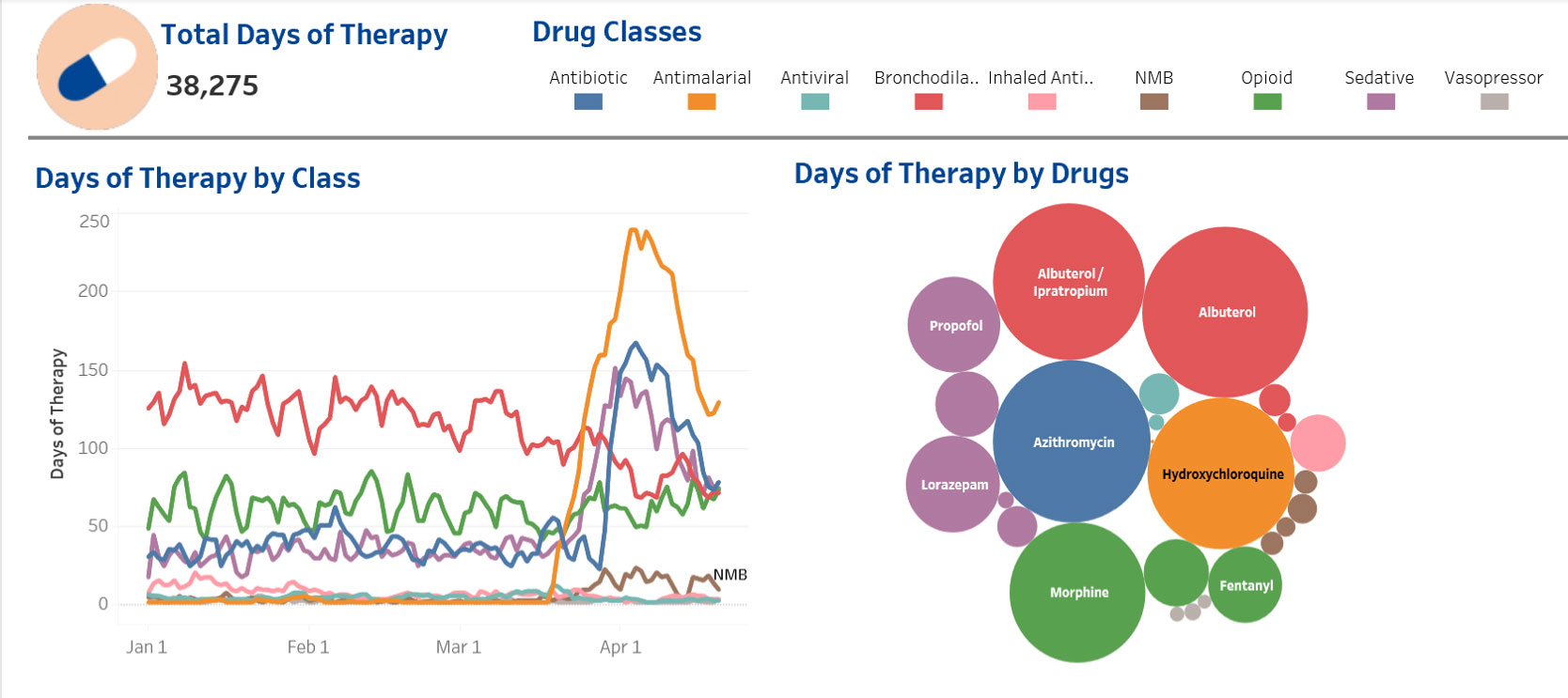
Oncology patients have complex medication requirements beyond their specific oncology regimen. Assessing the impact of interventions is made easier by data-analytics. ‘Big data’ allows for rapid review of therapies across the organisation and by individual care-unit and helps guide us to do ‘the right thing in the right way’.
For organizations without extensive EHR penetration compounding connectivity can also be used to extend medication safety beyond the pharmacy and into the nursing unit via ‘lighter touch’ Bar Code Medication Administration (BCMA) Systems. Such solutions are less expensive than full EHR bidirectional interoperability and can work alongside solutions with all the advantages of compounding software to smooth the workflow of production, married to barcode-supported right patient-right medication verification. It is possible to use this as a pit-stop on the journey to full EHR-interoperability. Medication safety should always be an ongoing journey to zero patient harm and scalability and adaptability of solutions. ‘Futureproofing’ is a vital consideration for organizations when building their IV-medication risk management strategies.
The ‘light touch’ solution requires less infrastructure and complexity as it can stand outside of the EHR or have touchpoint integration only, such as access to Admission Discharge and Transfer (ADT) data from the EHR.
Workflow for integrated CPOE to compounding with bedside BCMA scanning by nursing staff to support right patient-right medication verification at the bedside supported by a compounding solution:
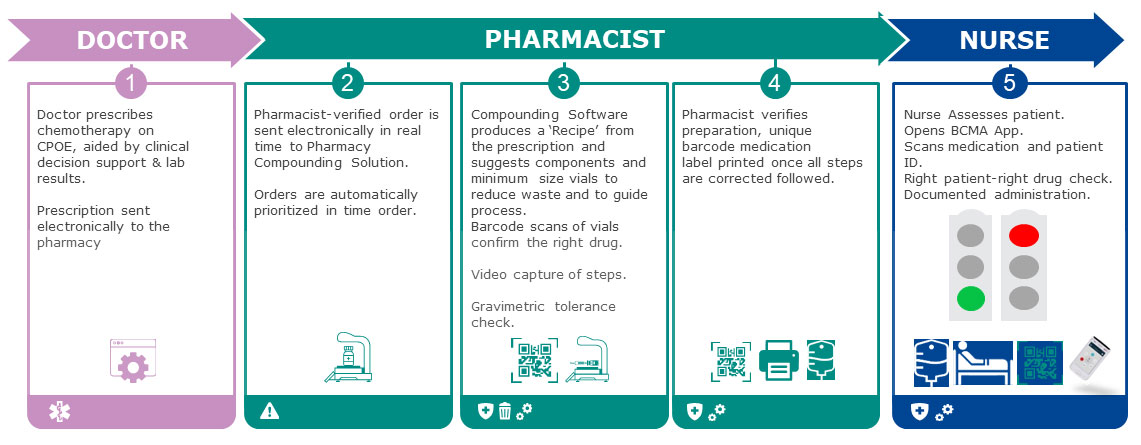
This requires investment in wireless connected handheld barcode scanning devices or the deployment of a proprietary application to nurses’ smartphones, but these scanning devices and applications have many other functions for nurses in terms of scanning for a positive-patient-ID for laboratory studies, oral medication administration, and for documenting patient transfers between units.
In the light BCMA solution the patient ID band and the patient-medication barcode label on each compounded product are scanned and this potential match is transmitted to a listening-post interface and through to the compounding solution server, where the match is checked. If it is correct for patient, medication, dose, volume and regimen appropriateness (oncology regimens, of course, contain multiple medications and are timed and scheduled as per the CPOE order) the nurse gets a green light on the bedside scanning device, and can proceed with administration.
This workflow achieves several things. It replaces a large amount of manual activity by nursing staff and should reduce the risk of medication errors as it undertakes positive patient ID and matching of the compounded product to the patient. It also means this activity takes place directly at the bedside, avoiding the risk of checks being made at the nursing station and then a mix up occurring due to distraction or other error at the patient’s bedside. The two-nurse or ‘four-eyes’ check is also not required as the barcode acts to verify fully, and of course there are many reports of the ineffectiveness, dangers of positive reinforcement of error, and of time-wasting that occurs when verification is undertaken manually by two clinicians. [11,13] Printed pages for regimens, which can be mislaid or placed in the wrong patient record, are also eliminated in the BCMA approach.
As discussed above safety systems can also streamline practice. In a recent study in a high turnover oncology unit with 16,000 patient visits and over 14,000 compounded medications produced per annum the introduction of a compounding software solution with BCMA labelling saw a 35% reduction in the time required to compound each product. The introduction of handheld barcode scanners for patient-medication verification led to patient verification time falling from ≈ 6 minutes to a mean average of only 41 seconds within two weeks of its introduction. When calculated against the checking of 14,000 medications. This gave a saving of nursing time equivalent to 0.43 fulltime nurses per year. [7] The savings for a 1,000-bed hospital compounding 40,000 oncology medications per year would be 1.21 full time nurses.
Such savings are vital as nursing staff shortages are now chronic and worsening, particularly in specialist areas like oncology. The world is ‘short of’ 30·6 million nurses and midwives and the Middle East has one of the lowest Human Resources for Health (HRH) densities for nurses, physicians and pharmacists globally. [14]
Technology cannot do everything that health care staff can do, but what it can do is release our most valuable asset, our highly skilled pharmacists, oncologists and oncology nurses, to undertake patient work of real value and to allow them to work in an environment that supports them in keeping patients safe.
References:
- Stanhope N, Vincent C, Taylor-Adams S, O’Connor A, Beard R. Applying human
factors methods to clinical risk management in obstetrics. Br J Obstet Gynaecol 1997 Nov;104(11):1225-32.
- Quinn C. Smart practice: the introduction of a dose error reduction system. Br J Nurs. 2011. 20(8 Suppl): S20–5
- Maddox R, Danello S, Williams C, Fields M. Intravenous Infusion Safety Initiative: Collaboration, Evidence-Based Best Practices, and “Smart” Technology Help Avert High-Risk Adverse Drug Events and Improve Patient Outcomes. In: Henriksen K, Battles JB, Keyes MA, Grady ML, editors. Advances in Patient Safety: New Directions and Alternative Approaches (Vol. 4: Technology and Medication Safety). Rockville (MD): Agency for Healthcare Research and Quality (US); 2008 Aug. PMID: 21249948.
- Waterson J. Making Smart Pumps smarter, making IV-therapy safer. British Journal of Nursing 22. 2013: 22-27.
- Biltoft J, Finneman L. Clinical and financial effects of Smart Pump-electronic medical record interoperability at a hospital in a regional health system. Am J Health Syst Pharm 2018 Jul 15;75(14):1064-1068.
- Wilson B, Jacob S, Yap M, Ferlay J, Bray F, Barton M. Estimates of global chemotherapy demands and corresponding physician workforce requirements for 2018 and 2040: a population-based study. May 8, 2019. Lancet.
- Meren ÜH, Waterson J. Evaluating an Automated Compounding Workflow Software for Safety and Efficiency: Implementation Study. JMIR Hum Factors 2021;8(4):e29180
- Reece K, Lozano M, Roux R, Spivey S. Implementation and evaluation of a gravimetric i.v. workflow software system in an oncology ambulatory care pharmacy. Am J Health Syst Pharm 2016 Feb 01;73(3):165-173.
- South DA, Skelley JW, Dang M, Woolley T. Near-miss transcription errors: a comparison of reporting rates between a novel error-reporting mechanism and a current formal reporting system. Hosp Pharm 2015 Feb;50(2):118-124.
- Liu J. The history of monoclonal antibody development – Progress, remaining challenges and future innovations. Ann Med Surg (Lond). 2014 Sep 11;3(4):113-6.
- Waterson J, Al-Jaber R, Kassab T, Al-Jazairi AS. Twelve-Month Review of Infusion Pump Near-Miss Medication and Dose Selection Errors and User-Initiated “Good Save” Corrections: Retrospective Study. JMIR Hum Factors. 2020 Aug 11;7(3).
- Terkola R, Czejka M, Bérubé J. Evaluation of real-time data obtained from gravimetric preparation of antineoplastic agents shows medication errors with possible critical therapeutic impact: Results of a large-scale, multicentre, multinational, retrospective study. J Clin Pharm Ther 2017 Aug 21;42(4):446-453.
- Schwappach D, Pfeiffer Y, Taxis K. Medication double-checking procedures in clinical practice: a cross-sectional survey of oncology nurses’ experiences. BMJ Open. 2016;6(6).
- GBD 2019 Human Resources for Health Collaborators. Measuring the availability of human resources for health and its relationship to universal health coverage for 204 countries and territories from 1990 to 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2022 Jun 4;399(10341):2129-2154.